February 17, 2026
Uncategorized
Defective Products & Drugs

Currently, there are numerous cases where individuals are suffering due to faulty products or drugs. Please review the list below. If you, or someone you know has been affected, please contact us immediately.
Cardiac Heater/Cooler Device
- Used during cardiac surgery to maintain blood temperature, sterilization procedures did not completely clean the device leading to infection
- Manufacturer: Sorin / LivaNova
- Damages: Can cause a rare slow growing infection with Non-Tuberculosis Mycobacteria (NTM)
- Case Qualification: Must have had surgery in the last five (5) years, must have notice from the facility of exposure to the device, and a diagnosed Non-Tuberculosis Mycobacterial infection.

Concerta
- Used to treat Attention Deficit Hyperactivity Disorder (ADHD) & Attention Deficit Disorder (ADD)
- Manufacturer: Janssen
- Damages: Suicide and severe injuries from attempted suicide
- Case Qualification: Suicide or injuries from a child 13 years of age or younger recently prescribed Concerta

Essure
- Essure is form of implantable permanent birth control for women
- Manufacturer: Bayer
- Damages: Painful complications leading to Hysterectomy, partial hysterectomy, product removal, migration, nickel allergy
- Case Qualification: Patient must have undergone a hysterectomy, partial hysterectomy, or product removal, or have suffered from product migration. Cases where the product is still in place with no issue or allergy-only cases will not qualify at this time.

Hernia Mesh Complications
- Mesh is used to repair a weak spot or tear in abdominal muscle (hernia). Composite mesh is made up of several layers of different materials.
- Manufacturer: Johnson & Johnson subsidiary: Ethicon, Atrium Medical, C.R. Bard subsidiary: Bard Davol; Medtronic (formerly Covidien) & others
- Damages: Infection, severe pain, hernia recurrence, adhesions, organ damage, product migration
- Case Qualification: Product must be a composite mesh implanted after 2009, with revision or scheduled revision, severe pain, bowel blockage, adhesions, infections. Priority Manufacturers are Bard, Covidien, Atrium, & Ethicon

High Blood Pressure Medications: Valsartan, Losartan, Irbesartan
- Drugs are prescribed for high blood pressure. Changes in the manufacturing process led to a large number of batches being tainted with NDMA, an extremely carcinogenic compound.
- Manufacturer: Various Generic Manufacturers
- Damages: Acute liver injury or cancer after ingesting valsartan or combination medication tainted with NDMA
- Case Qualification: Diagnosis of cancer of the liver, kidney, intestine (including colon and rectal cancer), or stomach after ingesting tainted valsartan (or combo drug) for at least one (1) year prior starting 2014 or later.
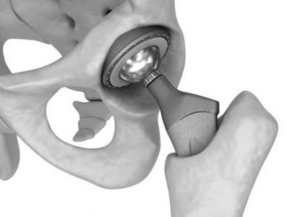
Hips – Metal on Metal or Recalled Devices
- Product is used as a hip replacement
- Manufacturer: DePuy & Stryker
- Damages: product failure resulting in revision or repair surgery, or notification of recall resulting in replacement
- Case Qualification: Product must be a recalled device implanted from 2007-2016, preferred cases have a positive cobalt test or are revised Stryker devices.
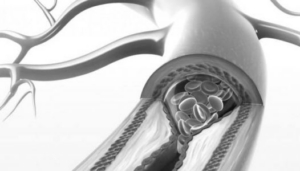
IVC Filter
- Product prevents blood clots and pulmonary embolism
- Manufacturer: CR Bard/BPV & Cook Medical
- Damages: Product Migration, perforation of organs, fractures, or post implant thrombosis
- Case Qualification: Any device implanted from 2003 to present day.
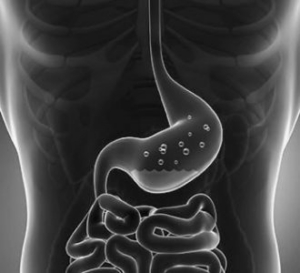
Proton Pump Inhibitors: Nexium, Prilosec, Prevacid
- Drugs are prescribed to treat persistent heartburn and acid reflux disease
- Manufacturer: Various Manufacturers
- Damages: Chronic kidney disease, acute kidney injury, such as interstitial nephritis, (NOTE: The following injuries have also been alleged, but we are not accepting these cases: dementia, bone fractures, cancer, and increased risk of heart attack)
- Case Qualification: Brand name or OTC use with related acute kidney injury (hospitalization is required) or diagnosis of chronic kidney disease Stage 3 or greater (1 year of use preferred); discontinuation of drug

Perfluoroalkyl Substances (PFAS)
- These substances include a number of chemically-related compounds used in industrial processes, manufacturing, and numerous household products, and are known to be carcinogenic.
- Manufacturer: Various Manufacturers
- Damages: Exposure can cause testicular and kidney cancer. Other disorders that have been attributed to, or associated with PFAS exposure, include high cholesterol; pre-eclampsia(also known as toxemia, a potentially dangerous complication of pregnancy); thyroid dysfunction (particularly in young children) and cancer; mental impairment and developmental disability; infertility; autoimmune disease; blood disorders; ulcerative colitis

Roundup® (glyphosate)
- Product is used in commercial and residential applications for the control of weed.
- Manufacturer: Monsanto
- Damages: Potential for various cancers, priority cases are Non-Hodgkin’s Lymphoma
- Case Qualification: Individuals who have experienced significant exposure to Roundup®, and have been diagnosed with a form of B-cell or T-Cell non-Hodgkin’s Lymphoma, including its many subtypes such as follicular lymphoma, chronic lymphocytic leukemia, mantle cell lymphoma, diffuse large B-cell lymphoma, and hairy cell leukemia.

Talcum Powder
- Talcum powder was marketed for feminine hygiene, and later found to cause the development of a variety of cancers.
- Manufacturer: Johnson & Johnson
- Damages: ovarian cancers, fallopian tube cancers, or mesothelioma
- Case Qualification: Ovarian cancer, fallopian cancers, or mesothelioma diagnosed by the age of 70; consistent use of talcum powder for at least five (5) years; negative BRCA 1 genetic testing (if tested), exposure post 1982, and cancer diagnosis preferably 2000 or later (we will evaluate all meso cases)

Textured Breast Implants
- For Illness related to a rupture, only the Mentor® Brand is accepted, and only if installed after 2006.
- For cancer cases, cases are specific to Non-Hodgkin’s Lymphoma.

3M Dual Combat Arms Earplugs
- Double ended ear plugs to prevent hearing loss during combat situations
- Manufacturer: 3M
- Damages: Allowed hearing loss to occur
- Case Qualification: Use of “3M Dual Ended Combat Arms Earplugs” (yellow and dark green), issued from 2003-2015, and client must have been discharged prior to 1/2015 and currently receiving VA benefits.

Valsartan
- Blood pressure and heart medication
- Manufacturer(s): Actavis; A-S Medication Solutions; AvKARE; Bryant Rank Prepack; Camber Pharmaceuticals; Hetero Labs, Inc.; HJ Harkins Company; Major Pharmaceuticals; Mylan Pharmaceuticals; Northwind Pharmaceuticals; NuCare Pharmaceuticals; Preferred Pharmaceuticals; Prinston Pharmaceutical; RemedyRepack Inc.; Solco Healthcare; Teva Pharmaceuticals; and Torrent Pharmaceuticals Limited.
- Damages: Cancer-causing substances N-nitrosodimethylamine (NDMA), N-Nitrosodiethylamine (NDEA), and/or N-Methylnitrosobutyric acid (NMBA) resulting in liver, kidney, stomach, or intestinal cancer.

Zantac
- Zantac can be contaminated with the cancer-causing substance N-nitrosodimethylamine (NDMA).
- Manufacturer: Sanofi
- Damages: In addition to stomach cancers, there are concerns that Zantac, ranitidine, and the associated presence of NDMA may also cause colorectal cancers or possible bladder cancers.
Zostavax
- Vaccine, intended to prevent shingles
- Manufacturer: Merck
- Damages: Causes shingles. Patients who are vaccinated with Zostavax may also experience one or more of the following injuries: necrotizing retinitis; hematoma (unusual bruising and swelling); itchiness (pruritis); muscle and joint pain; pulmonary edema (fluid buildup around the lungs)


