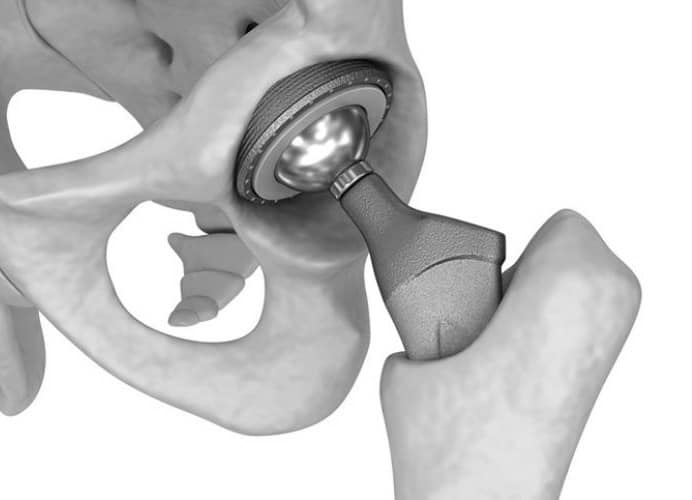
Hips – Metal on Metal or Recalled Devices
- Product is used as a hip replacement
- Manufacturer: DePuy & Stryker
- Damages: product failure resulting in revision or repair surgery, or notification of recall resulting in replacement
- Case Qualification: Product must be a recalled device implanted from 2007-2016, preferred cases have a positive cobalt test or are revised Stryker devices.
Meet Our Attorneys
The Aloia Roland practice areas are shaped by our attorneys’ diverse experience. Led by a talented team of trial attorneys, we function as a full-service law office and cater to our clients needs.

Evan D. Lubell

Jack C. Morgan III


